> Habitat & Distribution
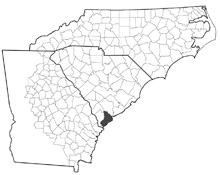
The native range of Pyrgophorus parvulus is the Caribbean rim and coastal South America including Cuba, the Lesser Antilles, Venezuela, and Mexico (Harrison 1984). Populations inhabiting coastal regions of southern Florida were described as Pyrgophorus platyrachis by Thompson (1968). The single population of which we are aware in southern Atlantic drainages, inhabiting a brackish system of ditches and drainage ponds on Hilton Head Island (SC) has probably originated from an artificial introduction. Pyrgophorus parvulus is pseudo-rare in North America, FWGNA incidence rank I-1p.
> Ecology & Life History
A wealth of detailed habitat data are available for P. parvulus populations inhabiting the Lesser Antillean Islands of St. Vincent (Harrison & Rankin 1978) and St. Lucia (McKillop & Harrison
 1980, McKillop et al. 1981). On St. Vincent, the maximum population density (766/m2) was recorded on mud and organic debris at the estuarine opening of a coastal rivulet. Dasheen marshes (Colocasia esculenta, or Taro) were the preferred habitats on St. Lucia, with population densities sometimes exceeding 15,000/m2 at peak reproduction in the fall. The introduced population on Hilton Head Island seems to reach (much lower) maximum densities on dead leaves, sticks, and organic debris in sandy ditches at a (surprisingly high) salinity of 14.5 ppt, and extends into salinities as high as 17.9 ppt.
1980, McKillop et al. 1981). On St. Vincent, the maximum population density (766/m2) was recorded on mud and organic debris at the estuarine opening of a coastal rivulet. Dasheen marshes (Colocasia esculenta, or Taro) were the preferred habitats on St. Lucia, with population densities sometimes exceeding 15,000/m2 at peak reproduction in the fall. The introduced population on Hilton Head Island seems to reach (much lower) maximum densities on dead leaves, sticks, and organic debris in sandy ditches at a (surprisingly high) salinity of 14.5 ppt, and extends into salinities as high as 17.9 ppt.
Pyrgophorus is an ovoviviparous brooder, females holding eggs until hatching in an modified oviduct sometimes called a brood pouch or uterus. The St. Lucia populations studied by McKillop and colleagues showed continuous base-line reproduction throughout the year and little cohort structure, making generalizations about life history difficult. But maturity seems to be reached in about four months, with peak reproduction August-October, and a second reproductive peak sometimes observable in the spring.
> Taxonomy & Systematics
Pyrgophorus populations often demonstrate striking polymorphism in shell morphology. The Hilton Head population includes animals bearing both smooth shells and carinate or spiny shells, varying in their body whorl size and spire height as well (see photo below). This has led (rather predictably) to the description of more than 40 nominal species and subspecies of Pyrgophorus. Hershler & Thompson (1992) wrote, We are uncertain if more than just a few of these are valid or if only one should be recognized.
Guilding described his Paludina parvula from the Caribbean island of St. Vincent in 1828. Ancey (1888) proposed Pyrgophorus to include Guilding s parvulus and many other nominal species of hydrobiids bearing spiny shells (e.g. coronatus Pfeiffer 1840, spinosus Call & Pilsbry 1886). But the biological similarities between the New World Pyrgophorus and the worldwide invasive Potamopyrgus (Stimpson 1865) are striking. Pilsbry and many other authors referred parvula to Potamopyrgus, rather than to Pyrgophorus, through the late 19th century and most of the 20th.
Pyrgophorus is classified in the hydrobioid family Cochliopidae, its distinctive penis fringed with numerous papillae (Hershler & Thompson 1992). A molecular phylogenetic study of the group has been contributed by Liu and colleagues (2001).
> Maps and Supplementary Resources
- Pyrgophorus distribution in Atlantic drainages (2023)
- A sample from the Hilton Head P. parvulus population, showing shell variety.

> Essays
- I reported discovery of the South Carolina Pyrgophorus population in my essay of 16Dec15, The Many Invasions of Hilton Head.
- Earlier versions of this website, online until August of 2016, adopted the large, broadly-inclusive concept of the Hydrobiidae (sl) following Kabat & Hershler (1993). More recently the FWGNA project has shifted to the Wilke et al. (2013) classification system, distinguishing a much smaller Hydrobiidae (ss) and elevating many hydrobioid taxa previously ranked as subfamilies to the full family level. For more details, see The Classification of the Hydrobioids.
> References
Ancey, M. C. F. (1888) Etude monographiques ur le genre Pyrgulopsis. Bulletin de Societe malacologique du France 5: 185-202.
Guilding, L. (1828) Observations on the Zoology of the Caribbean Islands. Zoological Journal 3: 527-544.
Harrison, A. D. (1984) Redescription of Pyrgophorus parvulus (Gastropoda: Hydrobiidae) from St. Vincent, St. Lucia, and Grenada, West Indies. Proc. Acad. Natl. Sci. Phila. 136: 145-151.
Harrison, A. D. & J. J. Rankin (1978) Hydrobiological studies of Eastern Lesser Antillean Islands. III. St. Vincent: Freshwater mollusca-their distribution, population dynamics and biology. Archiv fur Hydrobiologie, Monographische Beitrage 54: 123-188.
Hershler, R. & F. G. Thompson (1992) A review of the aquatic gastropod subfamily Cochliopinae (Prosobranchia: Hydrobiidae). Malacological Review Supplement 5: 1 - 140.
Kabat, A.R., and R. Hershler (1993) The prosobranch snail family Hydrobiidae (Gastropoda: Rissooidea): review of classification and supraspecific taxa. Smithsonian Contributions to Zoology 547:1-94.
Liu, Hsiu-Ping, R. Hershler & F. G. Thompson (2001) Phylogenetic relationships of the Cochliopinae (Rissooidea: Hydrobiidae): an enigmatic group of aquatic gastropods. Molecular Phylogenetics and Evolution, 21(1): 17-25.
McKillop, W. B. & A. D. Harrison (1980) Hydrobiological studies of Eastern Lesser Antillean Islands. V. St. Lucia: Freshwater habitats, water chemistry and distribution of freshwater molluscs. Archiv fur Hydrobiologie Monographische Beitrage 57: 251-290.
McKillop, W. B. & A. D. Harrison (1982) Hydrobiological studies of Eastern Lesser Antillean Islands. VII. St. Lucia: Behavioural drift and other movements of freshwater marsh molluscs. Archiv fur Hydrobiologie 94: 53-69.
McKillop, W. B, A. D. Harrison & J. J. Rankin (1981) Hydrobiological studies of Eastern Lesser Antillean Islands. VI. St. Lucia: Freshwater molluscs and the marsh environment. Archiv fur Hydrobiologie, Monographische Beitrage 58: 357-419.
Thompson, F. G. (1968) The Aquatic Snails of the Family Hydrobiidae of Peninsular Florida. Gainesville: University of Florida Press. 268 pp.
Wilke T., Haase M., Hershler R., Liu H-P., Misof B., Ponder W. (2013) Pushing short DNA fragments to the limit: Phylogenetic relationships of hydrobioid gastropods (Caenogastropoda: Rissooidea). Molecular Phylogenetics and Evolution 66: 715 736.








